A potato battery is an interesting way to introduce kids to basic electricity. They are cheap, easy-to-make, safe experiments that will teach them about simple circuits. This experiment is an easy way to teach kids about basic electricity.
Purpose: The purpose of the experiment is to show how you can use potatoes as a power source.
Check out our Lemon Battery Experiment

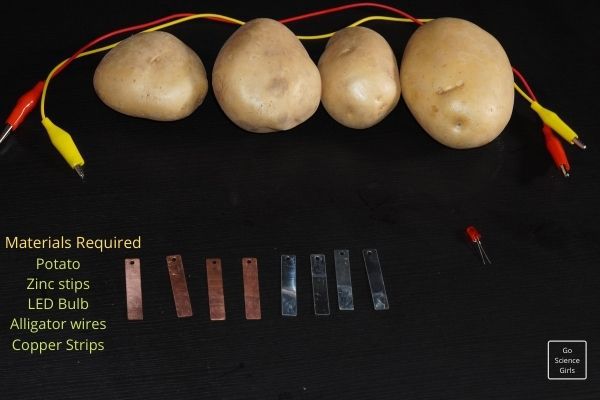
Materials Needed
Get these handy before starting.
- 2 large potatoes
- 2 zinc nails
- 2-3 pieces of copper wire
- 1 light bulb or LED light
- Tube of Glue
Alternatively, you can also use ready to use kits :
Building a Potato Battery – Procedure
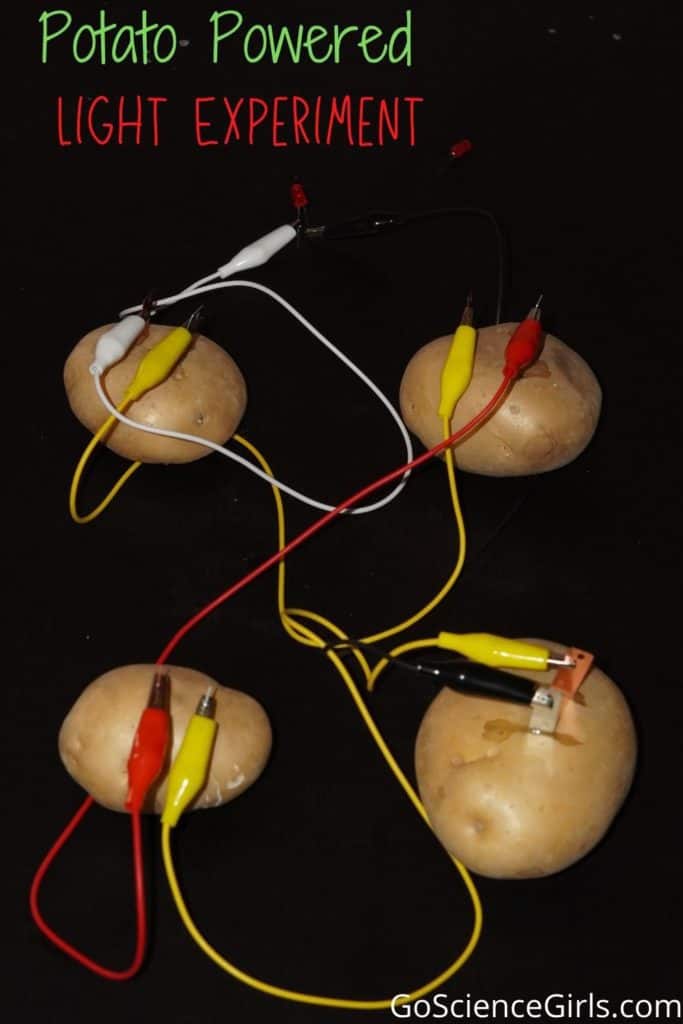
- Place the large potatoes on your experiment desk/table. The reason why we chose this vegetable specific is that Potatoes have an acid in them that reacts with metals like copper and zinc.

2. Push the two zinc nails in both potatoes. One each.

3. After that, wrap one wire around either of the zinc nails. This is done because, during the chemical process, electrons start to flow from one electrode to the other.
They both have different charges, opposite to be precise since they are made of two different metals.
The potato does not have the ability to generate electricity on its own, it acts like a salt bridge connecting the positive and negative, the anode to the cathode. Thus, representing batteries.

4. Try wrapping or connecting the other wire with the remaining nail. This will close the circuit and ensure current flows.
Although two potatoes can generate electricity, they may not be able to power a huge device. For that reason, you could add as many potatoes and wires as you’d prefer. That would increase the voltage automatically.
Nonetheless, the procedure will remain the same and the overall effect will be provisional.

5. Connect both the copper wires to the bottom of the light bulb. Stabilize it on the desk so it sits upright. We’ll have one connection out of two established after this step is completed.
Result
The connected LED light bulb glows and when measured the voltage from the connecting wires. we found that when a new potato was used, the voltage was around 2 volts.
As we went on and used potatoes that were not as fresh, the voltage began to decline and eventually turned out to be almost zero volts.
How Does a Potato Battery Work
Have you really observed that certain batteries possess a positive copper side (represented by a plus [+] sign) as well as a negative copper end (represented by a minus [-] sign)? Zn is a material that enjoys sharing electrons with copper.
Normally, zinc just donates its electron to Cu metal and the reaction ends. You may create a loop with a continuous channel of energy by providing the electrons with a medium known as an electrolyte to aid them to migrate to the copper & a wire through which they can travel from the Cu back to the zinc.
A potato battery is a simple electrochemical cell that can be made from potatoes. This is an electrochemical cell that uses the rapid movement of negatively charged particles to transform chemical energy among two metallic electrodes.
The concentration of starch liquids in potato, coupled with the rods, enables the potato to work as a cell, according to the potato rechargeable batteries concept. Copper & zinc are indeed the metals utilised in this experiment, and they react with one another to generate chemical energy.
Copper atoms are more attractive to electrons than zinc atoms. Several electrons move from the Zn metal to the Cu when a piece of copper & a strip of zinc make contact. The electrons reject one another as they focus on the copper.
This movement of electrons stops whenever the force of repelling among electrons as well as the attraction of electrons on Cu are equalised.
Instead of generating electricity, the potatoes serve as just an electrolyte. As a result, by splitting zinc & copper, it compels electrons to pass via the potato, forming a closed loop.
A little portion of potato electrical energy is produced by only utilizing 2 potatoes. Its power output could be enhanced by increasing the count of potatoes.
Even though the 2 metals were not in contact without any of the potatoes, electrons might be transferred, but no energy would be generated because the loop will be incomplete.
Aside from carbohydrates, potatoes include a substantial quantity of electrolyte in the form of different soluble acids and salts.
The Zn wire inside the “battery” acts as a negative electrode called (anode), while the copper cable acts as a positive electrode called a cathode.
An oxidation and reduction reaction occurs on the anode and on the cathode respectively.
Chemical Equation for potato battery is :
Potato + Zinc ==> Zinc ion + Sodium ion
Potato Battery This reaction is a redox reaction, which means that the chemical reaction involves a change in oxidation state. In this case, zinc is reduced and sodium is oxidized.
The potato battery works by using zinc ions to reduce the sodium ions. These zinc ions then go on to oxidize the potato molecules and create electricity.
This chemical reaction occurs because of a change in oxidation state, which is why it’s called an electrochemical battery.
Potato Battery Kits
Now that we’ve got the scientific bit out of it, let’s move on to Potato Battery Kits you can find on Amazon. That way, you’ll be able to conduct the experiment at your convenience and ease, while remaining stress-free. Find the list below:
STEM Toys: Chemistry Engineering Lab

STEM toys will ensure your child gets to experience only the best!
It primarily fixates on the Potato Battery Charged Digital Clock, so you get to experiment on something other than an LED light. In addition to that, it’s child-friendly and makes the whole journey easy and fun for young enthusiasts by specifying each part of the process in a simple way. STEM also teaches its audience about the ‘transformative power of green science’ through their kit.
The Salt Water and Potato Battery Kit

Well, the exploration begins with this kit! it offers you several attributes you must always look out for in a kit. Firstly, it’s got 4 different types of output materials. While at home you’d be experimenting with an LED or tube light, this kit gives you the option to explore your ‘current’ with completely different sources. Trust us, it’s worth the money you’ll be spending.
Secondly, it is inclusive of extras. With 6 times the regular amount, the Salt Water and Potato Battery Kit gives you the same material with a higher quantity. Thus, you could conduct the same experiment six different times which is twice the number of times other kits would lend themselves to.
Thirdly, it lets you experiment with different inputs. Typically, we’d use a potato at home to conduct this scientific exploration, but this kit offers you other types of acids like lemon, vinegar, sodium etc. You’ll be exposed to so many more kinds of results and can compare them after you’re done playing around!
Lastly, the kit has a 10-page booklet that has instructions and other guidelines written down for you. That means you don’t have to go over the internet to understand what you’re supposed to do. It’s already there. But hey, don’t forget to read our post!
Get yours as quickly as possible!
We are in love with these kits. They have something for everyone! We hope you all had a great time learning about the experiment and wish you all the best in your own science experiments!
What to do in case the experiment fails?
There are times you might not be successful in generating electricity. This could be due to the following reasons:
- Loose Nails: You need to ensure that nails are properly inserted into the potato.
- Loose Wires: Make sure to wrap your wires around the nails tightly. At times, they start to de-coil. This will hamper the experiment as there will be no flow of current.
- Type of metal: You must understand that potatoes react with specific metals only. Another property to keep in mind is that the metals need to oppose each other.
- Condition of potato: Your chosen potatoes must exist in their natural conditions. You have to do nothing to them. Bare potatoes will work most efficiently.
Safety Measures you must take
- All experiments require adult supervision.
- Please don’t conduct the experiment if you are not familiar with the process. This is a very easy and fun science experiment for kids of all ages, but can be dangerous if performed incorrectly.
- Do not touch the battery terminals while performing this experiment.
- If you are not sure of something, please ask an adult before proceeding with the experiment.
FAQs
Besides these important aspects of the experiment, there are common questions that tend to arise. Let’s try looking at a few:
While this is one of the most frequently asked questions, the answer still remains subjective. There are several factors that are at play during the experiment. Reducing or increasing the effect of one on the other will tamper with the final results.
Generally, one potato produces around 0.5 volts of energy. However, a boiled one can produce 5.
If you’re stranded on an island with no battery on your phone, but a potato in your backpack, chances are you could be saved.
It’s simple math. 1 potato generates 0.5 volts and 0.2 milliamps. Your phone charger output requires 5 volts and 2 milliamps to begin charging your phone. Thus, 10 potatoes would work perfectly. However, if you’re at home and have the patience to boil a potato, you’d only need 1-2.
Even a single potato could do wonders for you. The only things you must keep in mind is the acid in the potato and the condition of the nails and wires. They shouldn’t corrode. If that’s sorted, then your battery could last for several hours. Some people even run it for days. That’s because they have good quality, stable equipment at their disposal.
