In this article, lets explore how balloon with water does not burst when exposed to fire.
This Fireproof Balloon Experiment can help kids to learn how heat gets conducted through different materials. This is a great activity to teach all about thermal conductivity.
In this simple science activity, children can hold the balloon over the fire without popping it.
Fireproof Balloon Experiment
The experiment helps kids to understand more about the difference between theremal conductivity of water and air. In addition, they also learn about the transfer of heat in air vs. water.

Things we need
1) Balloon
2) Water
3) Any heat source that emits fire like candle, matches, or lighter
4) Safety Goggles (optional)

That’s it! Just collect these materials and get ready to experience the mind-blowing science of balloons, water, and heat.
Before we land on the experiment directions, let us discuss a few questions that help to awaken your child’s predictions on the activity!
1) What happens to the balloon when brought closer to the fire?
2) What is the role of water inside the balloon?
3) Why the balloon with water does not burst over a hot flame?
4) What is more conductive between water and air?
5) Define the black soot formed on the balloon after the activity.
Simple Step by Step Instructions to make The Fireproof Balloon
Step-1: As a first step, blow up the balloon in the same way as you regularly do and give it a tight knot at its mouth part.
Step-2: In the second step, light up the candle and place it on the experiment table. And bring the inflated balloon as closer as possible over the flame. The moment the balloon experiences the heat, it pops out.

Note: Do not forget to direct your child to put on their safety glasses and hand gloves as they are dealing with fire.
Repeat the same experiment but this time using a little bit of water.

Step-3:
- In this step, ask your child to blow up another balloon to the same size as the first balloon.
- Add a tiny amount of water into it to form a layer at the bottom of the balloon.
- Later, please give it a knot as usual.
Step-4: As a fourth step, bring the inflated balloon filled with water over the flame-like just 3-4 cm away from the fire. Whatever the angle you hold the balloon, you need to make sure that the water layer touching side of the balloon is upon the flame.
Step-5: The final step is to observe the outcome! The inflated balloon with water inside does not pop out even if you make it touch the fire. This is the outcome you must witness after the experiment.
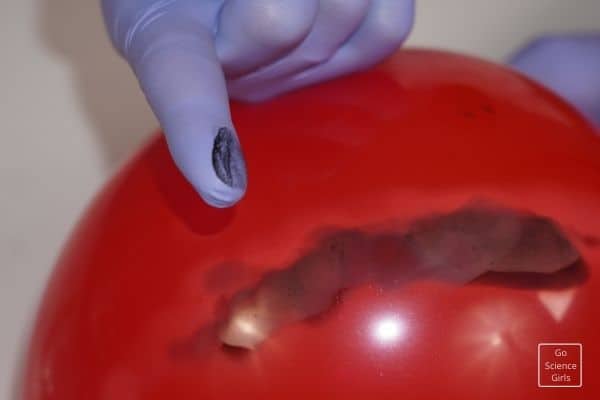
Note: If your balloon blows out even after filling with water, you are going wrong in holding the balloon in the right place. So, re-check the instructions and experiment to see our fire-resistant water balloon.
Here is a small step to do, i.e., continue to hold the balloon over the flame. You will see a black soot formation on the balloon at the point of contact with the fire. To know why to continue reading to explore the science behind it.
The Science behind the Fireproof Balloon Experiment
Materials when exposed to flame/fire, they catch fire only when their temperature reaches above the flash point.
Flash point is nothing but the temperature at which the material itself catches fires and burs on its own.
When the balloon with water exposed to flame, the latex material gets heated up on one side. However since we have water on the other side of material, the heat gets absorbed by the water and the water temperature rises instead of the latex’s temperature.
Water is a great conductor of heat and absorbs heat much better than air. That is the reason the water absorbs heat and changes its temperature when brought over the flame.
On the other hand, the latex material of the balloon also supports the transfer of heat through and inside it.
The heated water molecules have less density and travel upwards, i.e., towards the cooler side. In contrast, the space left by hot water molecules substituted by the cold water molecules. And these cold water molecules get heated up and travel upwards again. So this process inside the balloon continues as a cycle.
As the cycle of exchanging water continues, it makes the balloon fire-resistant and does not allow the balloon to blow up! The balloon can hold the heat until the heat of a fire is greater than the water conductivity of heat.
So, all the magic of resisting heat by the balloon lies in the heat conduction process by water.
The soot formation at the end of the activity is from the flames coming out of the candle’s fire. That is nothing but carbon, the primary substance present in the heat flames. It forms as a layer of black coating on the balloon and keeps it undamaged.
Here is a list of interesting ballon activities to try at home:

Discussion (extension) Ideas after the experiment
1) In what way, our demonstration relates the temperatures over ocean water and the coastal areas?
2) Will there be any change in the experiment results if you change the amount of water quantity inside the balloon?
3) Find out what the other liquids are the excellent conductors of heat! Perform the same experiment to learn the changes.
4) What is the reason behind the soot formation?
