Performing science experiments, especially by kids is so much fun. Hence to add more fun, we are here with another fun-loving experiment – How To Make Instant Ice?
Instant Ice formation has been proven beneficial in many aspects. Its endothermic nature and time-saving property have been immensely used in scenarios of cold-time crunch treatments. Instead of following those long processes, we can make it in a few seconds.
In this article, we have given step-by-step experimentation and detailed information regarding every single thing you need to know about making instant ice. So, go ahead and read below!
How To Make Instant Ice?
The most basic type of instant ice formation. No additional substances except water is used.
You may be interested in our Amazing 18+ Ice Science Experiments
Steps to make –
- Grab a dozen or so water bottles (better to be safe than sorry; if one bottle fails to show the desired product, another can be immediately used).
- Place them in the refrigerator for two hours (add a quarter or half-hour more, if the result is not achieved), preferably in a horizontal position.
- Gently take the bottles out on completion of the allotted time, and place them carefully on a flat surface.
For Instant Ice in a Bottle:
- Hold one of the cold bottles in your hand and slam it down hard on a flat surface.
- The bottle freezes rapidly from top to bottom (roughly 5-10 seconds), turning non-transparent and having your Instant Ice.
For Instant Ice as an Ice Sculpture:
- Pour the contents of one of the bottles on a piece of ice placed on a flat surface.
- As soon as the poured water comes in contact with the already ice placed, it transforms instantaneously. Continuous pouring causes the ice to accumulate in height, and an Instant Ice sculpture is created.
Instant Hot Ice Making
- In a beaker/pot, add 1 lt of baking soda slowly (to avoid a volcano, which may overflow) to 4 tbsp of vinegar with constant stirring. The following reaction occurs.
[HCO3]– + CH3–COOH → CH3–COO– Na+ + H2O + CO2
Sodium acetate and carbon dioxide are formed as products.
- The solution formed is a diluted one, and we need a concentrated one. To do so, boil the contents of the solution for about an hour or so until a thin film (100ml-150ml) is obtained. Do not worry about any discoloration that may occur.
- Remove the solution from the heat source and cover it immediately to protect it from any crystal formation. If crystals do appear, then mix a small amount of water until they dissolve.
- Place the solution into a refrigerator.
- For making “hot” ice sculptures, i.e., to occur crystallization, either touch the surface with an object or add a small preformed crystal of sodium acetate, which will set off the nucleation process.
- Pour the solution on a flat surface, and your “hot” ice sculptures are ready.
Science Behind Instant Ice Formation
The Experiment performed appears quite exceptional to the naked eye and indeed very awe-inspiring to those who do not have a science background.
Diving deeper into the ‘how’ and ‘why’ of it, we learn that this miracle is mind-blowing and has a very simple and straightforward explanation.
The mechanism behind the magical instant ice is a phenomenon called ‘Snap Freeze’.
The freezing point of water is 0℃ or 32℉. The technique of ‘Snap Freeze’ is to take water when it is super cool but hasn’t frozen yet; it’s just at the brink of it.
This method lowers the temperature to -70℃ or -94℉ in a “snap,” as the name suggests.
For the experiment to be successful, you must use supercooled water but should not reach the finish line of the freezing point yet.
On exposure of this “almost frozen water” to any kind of force, a slam or a whack (in case of instant ice in Bottle procedure) or to ice, which acts as a precursor to the pouring chilled water (in case of instant ice in Ice Sculpture procedure), ‘Nucleation’ occurs.
Nucleation
Nucleation is the changing of a substance from one state to another.
Water has numerous nuclei which act as a harbour for any impurity or the water molecule to turn into an ‘ice crystal’.
The ‘ice crystal’ initiates a chain reaction of crystal formations called ‘crystallization.’
A trigger for nucleation is a contact to any deformity. Hence, when slammed/whacked against a solid surface, it begins to freeze fast and within sometime the entire bottle is frozen.
Another trigger is, contact with already prepared ‘ice crystals’. Therefore, when it touches the ice, it freezes rapidly.
Both the cases are examples of ‘Heterogeneous Nucleation’.
Suppose, ice is formed naturally without any external disturbance or introduction of any impurity, just like the usual long-term method of storing water in the freezer for an extended period. In that case, it is an example of ‘Homogeneous Nucleation.’
Influencing Factors
- The volume of water.
- Dimensions of the bottle.
- Initial Temperature (before placing in the refrigerator) – preferably should be room temperature for the best outcome.
- The temperature of the refrigerator.
- Final Temperature (after placing in the refrigerator).
- Position of the bottle, since the more surface area exposed, the faster is the cooling process-
Vertical – less surface area exposed – slower cooling.
Horizontal – more surface area exposed- faster cooling.
Amount of impurities –
More – hinder cooling.
Less – smooth cooling.
Presence of the tiniest of impurities may set off preterm Nucleation.
Features/Conditions
- It can maintain its state as instant ice for about 15-20 seconds, variable, depending upon the environment it is being performed in and the climatic conditions of the location.
- Pure water is a compulsion for this experiment because being devoid of any impurities will lead the water to reach the freezing point before forming crystals accidentally.
- Alternatively, for an even quicker experiment time duration, add ‘salt’ to the pure water. The addition of ‘salt’ lowers the freezing point significantly. So, the refrigeration time can be reduced to 10-15 minutes which is remarkably lower than the standard period.
Liquid Nitrogen / Cryopreservation
‘Cryopreservation’ refers to preserving any substances, especially biological compounds, by subjecting them to lower temperatures. These usually range between -80℃ to -196℃.
Nitrogen’s freezing point is -210℃ or -346℉. It turns from gaseous to liquid state on attaining its freezing point and has an uncanny resemblance to water. The temperature of liquid nitrogen formed is -196℃ or -320.8℉.
This form of nitrogen applies to quick and rapid freezing, such as cooking competitions and bakeries, where time is of the essence.
The sample to be frozen is directly dipped into a container of any sort filled with liquid nitrogen. There is no chemical change in the substance subjected to this procedure.
Also, an added merit other than being a massive time-saver is, liquid nitrogen preserves the structure of the substance dipped in it and does not form crystals as seen in regular refrigerator freezing.
Purpose / Practical Applications of Instant Ice
1. Beverages
Since it is an ‘exothermic reaction,’ ‘latent heat’ is released in the ice as it freezes from top to bottom, heating it and increasing the water temperature.
The increase in temperature makes it a bit warmer, suitable for consumption. It can simply be taken or added to other beverages to enhance them.
E.g., It is added to fruit slushies like mango or strawberry to add a refreshing element to them.
2. Ice Sculptures
Different shaped ‘Ice Sculptures’ can be created by placing ice beforehand in the shape you wish to achieve. You will then simply have to pour the cooled water over the prepositioned ice, and your Ice Sculpture is ready!
3. Ice Packs
It is a 2-layered mobile bag which consists of – a plastic bag of water/water-laden gel, inside a bag of ‘Ammonium Nitrate, Calcium Ammonium Nitrate or Urea.
Here, the Ionic compound which is Ammonium Nitrate, Calcium Ammonium Nitrate or Urea, and the Non-Ionic compound is water.
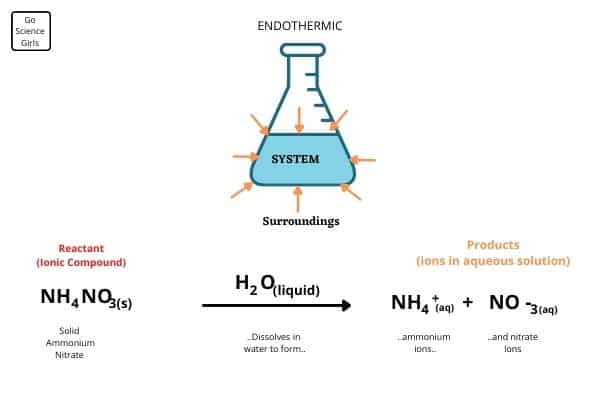
Interactions between an Ionic and Non-Ionic compound are always heat-absorbing.
When the barrier between the two bags is forcefully broken at the time of use, the contents of the two bags mix, and an endothermic reaction takes place, absorbing all the heat from the surroundings. Instantly, the bag turns cold, fulfilling its purpose.
The instant-mix-ready packs are crucial in treating primarily sports injuries or domestic accidents, like in sprains, strains, muscle pull, jaw injuries due to punches, etc.
4. Food Preservation
Delicate and perishable food items are subjected to sudden and instant ice to pause their physical and chemical processes, preserving their natural characteristics of taste and smell.
Their cell membranes remain intact even after denaturation. On heating, the food returns to its fresh form and is consumable.
Related Ice Experiments for you to try on:
Conclusion
Instant Ice is both exciting and valuable to work with. Its phenomenon is nothing short of miraculous.
Even though there is a scientific background to it, it will always be an astounding manifestation to witness.
For fun-loving science enthusiasts, coloured instant ice is the latest invention. It is done by adding colour-imparting pigments to the base mixture in both standard and hot instant ices.
It has spread its branches and roots of applications vastly in various aspects of provisions.
Be it food preservation, food preparation or an instant relief for an injured football player, instant ice has got everything covered.
Happy Ice-making!





