This science activity demonstration reveals the real science behind the strange phenomenon of dancing droplets.
Do you know droplets of liquids can also dance? Yes, they can dance well like performers instructed by a choreographer. Sounds unbelievable? Read on!
Materials Required
*Product Links are affiliate links – Your support is highly appreciated
Steps to be followed
- Take the sterilized glass slide and keep it on a flat surface.
- Next, deposit tiny droplets of different colors next to each other on the slide with the help of the dropper.
- Observe the actions.
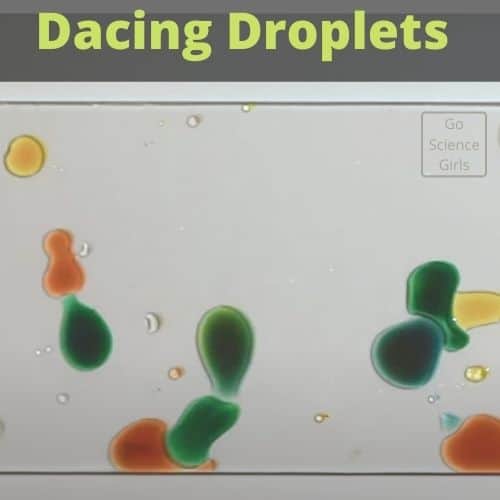
Science Behind Dancing Droplets
Courtesy: Stanford Physics Central
After you have done the short science activity, which is extremely easy, you will notice that these little droplets are moving as if they are sensing the presence of each other and chasing each other. This synchronized motion is both beautiful and amazing!
Now, you may be curious to know the science behind these dancing water droplets. You may don’t know that in most of the food coloring agents, there is a presence of propylene glycol along with water and other ingredients.
Now, propylene glycol is a synthetic liquid that absorbs water. It is the dynamic interaction of water and propylene glycol that makes these droplets dance but isn’t everything so confusing?
Let’s get it explained in a better way!
Actually, it is the evaporation and surface tension that makes these droplets dance. In each droplet, water is evaporating more quickly than propylene glycol, and water also has higher surface tension.
Here, while it is the evaporation that determines where the droplet should move on the glass slide, surface tension provides the necessary pull for the motion of these self-excited dancing droplets.

What is surface tension?
While you all must be well aware of the concept of evaporation, surface tension may not be something that you can understand. So, this is the tendency of liquid on a surface. Here, intermolecular forces of that liquid pull the liquid molecules together on that surface.
Do all liquids have surface tension?
Yes, all liquids have surface tension, but some have higher than the others. For example, water has a high surface tension.
However, its surface tension can be reduced with a bit of soap water. Check out the soap boat water experiment on our site to know how this all unfolds!
You will be surprised to know that the knowledge behind the phenomenon of dancing droplets can find its application to find new ways to clean glass surfaces or even self-cleaning solar panels!
In the video, you can also notice that how drawing back lines by using a sharpie pen on the glass slide and filling each basin with a different colored liquid makes the droplets to behave differently.
It seems that there are multiple catch basins on the glass slide, and the colorful droplets have found a mechanism to know which one has the color as they have!
So, just enjoy this super fun science activity at home and know more about such science secrets.

Resource
https://www.youtube.com/watch?v=ZMsaH6SY4CY&feature=emb_logo
https://news.stanford.edu/news/2015/march/dancing-droplets-prakash-031115.html
