Let’s discuss about ‘Balloon in hot and cold water experiment’ in this article. This interesting balloon experiment helps children to learn about density, surface tension, and air pressure.
Balloon in hot and cold water experiment
Hypothesis
- The volume of air changes based on the temperature surrounding it.
- Air expands or contracts based on increase or decrease in surrounding temperature.
Things you need to do Balloon Experiment
1) Two plastic/ glass container (bottles)
2) Balloons
3) Hot Water
4) Ice cubes as a cold water source
5) Containers to place hot and cold water

Preparation Steps
1) You can prepare your children or students by asking “How can you inflate the Balloon without touching it?”.
2) Note down their expected answers. But discuss their solutions only after performing the experiment to catch the science concepts behind it easily.
Step by Step Directions
Let’s start with the hot air experiment.
Step-1:
Take a glass container and add cold water. Then, add few ice cubes to it to keep it cold.

Step-2:
Pick another glass container and add some amount of hot water into it. Ensure the hot water’s hotness need not to be sizzling.

Step-3:
As step 3, bring our Balloon over the neck or mouth of the crystal clear plastic bottle in an upside-down position. And fix the mouth of the Balloon to the mouth of the bottle as shown in the picture.

Make sure the bottle is empty before you attach the Balloon to it.
Repeat the same method and prepare another set of water bottle and Balloon using the other empty bottle.
Step-4:
In this step, keep the ballon attached bottle inside the container, which consists of hot water. Let the bottle sit in hot water for some time.

You will observe the Balloon starts inflating itself without any external force. Amazing, isn’t it!?
Step-5:
And then bring the same and another set of water bottle into the container which consists of cold water. And allow it to sit for some time to see the results.

You will observe the Balloon starts shrinking itself by deflating the air inside it.

Note: If you feel the hot water is becoming cool, replace it with another hot water cup. In the same way, if you feel the cold water is becoming hot due to outside temperature impact, add some more ice cubes and make it cool. In this way, you can maintain the temperatures of the water while repeating the experiments.
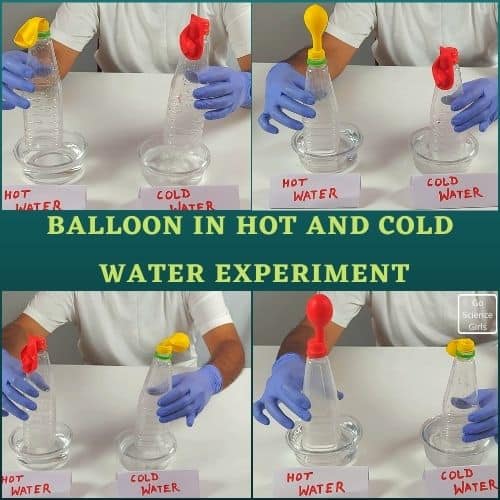
Science Behind Expanding Balloon on Hot Water
The quantity of air occupied in a particular space, i.e., an open or closed container, denotes ‘Volume.’
Well, an empty water bottle is also populated with a certain amount of air molecules inside it—the air molecules inside and outside the bottle move with equal pressures at normal surrounding conditions.
In this activity, when we attach a balloon over the bottle’s mouth and place it in a hot water container, the Balloon starts inflating. It is because the hot air molecules enter into the Balloon from the bottle, which is in a hot water container.
These hot air molecules move faster inside the Balloon and occupy more space as they become less dense than usual. When they become less dense, it requires more space to settle, and that is why the Balloon starts inflating to provide more space for hot air molecules.
And when the Balloon inflates in hot water, bring it into the container containing cold water. Here, the cold air molecules replace the hot air molecules because hot air molecules cool down due to cold water.
When the air molecules become colder, air molecules’ density gets back to a denser state and requires less space to occupy. That is why the inflated Balloon deflates when the bottle is placed inside a cold water container.
This is how the volume of air calculated:
Volume= Mass x Density

Safety Tips
Have adult supervision at all times during the experiment to avoid any unforeseen incidents.
Suggested to wear gloves and safety glasses while doing experiments with hot water.
Avoid handling hot water by small kids.
Learning for Elementary, Middle School, and High School Students
The same experiment can be used differently based on the level /grade of the students.
Elementary Students
When kids are in elementary school, it is the best time to learn about different states of matter, i.e., solids, liquids, and gases. Solids and liquids are visible to the naked eye, and hence students can easily catch up with the properties and characteristics. And it is easy for them to compare various objects and liquid things and determine the state of matter properties.
But when coming to gases, it is difficult for them to determine their properties because gases won’t appear to the naked eye, and children go confused. That is why we need to explain them clearly by concentrating much on performing various science experiments that involve gases. One such experiment is the ‘Balloon in a bottle’ experiment.
Through this experiment, students can quickly learn about gases and their properties.
Middle School Students
In middle school, students focus on macroscopic particles and determine the objects around them and tell whether they have solid or liquid or gaseous properties. Because at this level, they will get to learn about states of matter in regards to their arrangement, position, and movement. Also, they can explore that all forms of matter are made of atoms and molecules that consist of weight, especially gases. As the air is invisible, they think that gases do not have mass, but they learn about gases containing mass with this experiment.
Besides, they can explain the conservation of matter with a good reason using the concept of closed systems.
High School Students
At this level, as the name suggests, students become sharp and can apply their knowledge on gases. This knowledge helps in understanding even the difficult context of gases, i.e., ‘Gas Laws.’ Also, they can apply Charles Law and explain Gas Law. And using conservation of matter principles and laws, they will make out the differences in temperatures and their relation to the volume of gas.
In this way, students at different school grades learn the gaseous properties by performing this super classic experiment of ‘Balloon in a Bottle.’
Laws Behind the Experiment
Gas Law
Gas Law or Gas Laws is/are a collection of laws which include Boyle’s Law, Charles’s Law, Gay-Lussac’s Law, Ideal Gas Law, and Avogadro’s Law. These laws combine to state how an amount of gas reacts to changes in temperature, pressure, and temperature. The following are such statements these combined laws work on:
1) The complete temperature of a gas
2) The amount of volume working with a gas
3) The amount of pressure experienced between the walls of a container and a gas
4) The mass of a gas
The above-mentioned combination laws were a great invention during the 18th century, and here are the definitions of each law:
Boyle’s Law: The law which states the kith and kin between the volume and pressure of a given amount of gas is nothing but Boyle’s Law.
Charles’s Law: Charles’s Law is the law that tells about the absolute temperature of a gas and its association with the volume employed by it.
Avogadro’s Law: The type of law which states the correlation between the number of moles of a gas and the amount of volume occupied by it refers to Avogadro’s Law.
Gay-Lussac’s Law: Gay-Lussac’s Law tells that the relation between the absolute temperature and its pressure is directly proportional at constant volume.
Ideal Gas Law: Ideal gas law is a combination of three laws, i.e., Boyle’s Law, Charles’s Law, Avogadro’s Law, and hence refers to the term ‘combined gas law.’ This law states the differential behavior of gases at different conditions and concludes that a gas’s pressure is directly proportional to the absolute temperature.
Pressure, volume, and temperature are the three significant physical factors that determine the behavior of gases. When these parameters are at standard conditions, the activities of all types of gases remain the same. The states of gases can vary based on the condition.
So, the gas law and all other five laws state all gases’ behavior is associating with all three physical parameters.
Boyle’s Law Formula: P∝1/V
Charles’s Law Formula: V∝T
Avogadro’s Law Formula: V ∝ n
Ideal Gas Law Formula: PV= nRT
Gay-Lussac’s Law Formula: P ∝ T
Here, P= Pressure of the gas, V= Volume of the gas, T= Absolute Temperature of a gas, n= Number of moles, R= Equilibrium Constant.
Worksheets
Here are some worksheets that would complement the science experiment. Attempting these worksheets might help studnets to sustain the knowledge gained through the experiment. On the other hand, teachers use these worksheets to understand and monitor student’s previous and current knowledge.
https://scied.ucar.edu/sites/default/files/files/activity_files/BalloonOnBottle_0.pdf
https://www.flinnsci.com/api/library/Download/e2dfff9fc2324f51889429583a51ac63
https://ps21pd.weebly.com/uploads/1/2/0/6/12065719/kinetic_theory_-_hot_and_cold_balloons.pdf
Practical Applications
Let’s learn how to apply these science concepts in real life applications happening around us.
Hot air balloon: Yes, the science behind hot air balloon and Balloon in the bottle activities is similar, i.e., hot air rises, sending the cool air to replace the space created by it. When you provide heat flames in the hot air balloon set up, the heat energy enters into the Balloon.
Generally, the hot air consists of less dense air molecules, which tend to rise. That’s why and the hot air balloon rises in the sky until they provide enough heat.
Not only air, any substance that exhibits less dense molecules than the surrounding gaseous or liquid matters float. Forex: Wood floats on top of the water because wood consists of less dense molecules than water. This phenomenon of increasing the molecules’ speed regarding the increase in temperature of a gas refers to ‘Thermal Expansion.’ And the wonder of floating objects due to the pressure or force exerted is ‘Buoyancy.’
Sun Producing Wind on Earth: The winds produced by Sun on the Earth also exhibit the same phenomenon, i.e., thermal expansion and buoyancy.
Earth’s temperature is uncertain, so we cannot predict its long-term weather and climatic conditions. It is because different parts of Earth receive heat from Sunlight at different times as Earth is round and rotating.
So, the Sun can’t provide Sunlight to all parts of the Earth at the same time. Hence, Earth receives different air temperatures at places closer to the surface of the Earth. Besides, the Sun’s angle is focussing its Sunlight on the Earth also plays a significant role in changing the temperatures of Earth.
According to the above concepts, several continents on Earth receive more heat than other continents. Comparing land and water, land absorbs more heat faster than water, and therefore we see continents with more land exhibits high temperatures.
But during nights land releases heat more quickly than air and hence we feel cooler climates at night time. In this way, Earth reveals different climatic conditions and atmospheric temperatures during the day and night times.
Let us discuss these concepts in detail with a practical example, i.e., Off-shore and On-shore Winds. During nights, the oceans’ surface gets warmer so quickly because the surrounding land cools down and shows lesser temperatures.
As a result, the warmer air becomes less dense and rises upwards, leaving the space on the surface occupied by the cold air from the land. Thus, creating the off-shore winds that produce renewable and pure energy.
And at daytime, we experience on-shore winds that mean the land absorbs more heat from the Sun and exhibits warmer air. This hot air does not remain on the land surface; instead, it rises into the air because it consists of less dense air molecules.
Simultaneously, the temperature at the ocean level exhibits less heat than the land surface temperature. So, the cold air from the ocean surface replaces the hot air molecules’ space creating on-shore winds.
Lesson Plan
Here is the best lesson plan on the ‘Balloon in hot and cold water’ experiment.
Preparations
1) Ask the students whether they can inflate the Balloon without touching it. Note down their answers and discuss their solutions after the experiment.
2) First, invite your student’s answers and discuss their solutions with a scientific reason.
3) You can encourage and inspire students by telling them that they are upcoming engineers, chemists, and other respectable designations. Forex: if a student predicts the answer would be ‘by adding baking soda and vinegar,’ explain why his response went wrong. Then, encourage him by saying he/she is thinking smartly like a chemist. In this way, depending on their predictions, a teacher can inspire them with specific designations.
4) If a student does not respond to your challenge of inflating a balloon without touching it, then give him an example and ask him/her to compare. Let the student come up with his/her answer with a bit of explanation.
Directions
Guide your students on the instructions of the ‘Balloon in hot and cold water’ experiment step by step, clearly as mentioned at the top of this post. You can also ask and discuss a few questions related to the subject while experimenting. Such that students feel more encouraged and involved in the topic rather than feeling bored.
Here are the basic questions you can discuss with students:
1) Why does the Balloon inflated on itself?
2) What is the difference between hot and cold water changes and their impact on the Balloon?
3) How long the Balloon takes time to inflate itself in hot water?
Explain about Misconceptions
Students think that hot air blows up the Balloon as the hot air rises upwards. But prove it as a misconception by reversing the bottle with an inflated balloon. Still, the Balloon remains inflated without deflating. It is because hot air rises when there is cold air beside it.
Finally, explain the background science involved in this experiment and discuss students’ predicted answers with a scientific reason. Tell them clearly that their answers may not apply in this science activity, but they may use them in another way of experimenting.
FAQ’S
In hot water, the Balloon inflated because of hot air molecules, and in cold water, the Balloon deflated because of cold air molecules. The hot air molecules are less dense in weight and tend to rise and occupy more space. That’s the reason the hot air molecules travel inside the Balloon and make it expand. In contrast, the cold air molecules are denser in weight and require less space, causing the Balloon to deflate.
Take an empty plastic water bottle. Attach a balloon (make sure it is not leaking anywhere on its surface) to the bottle’s mouth using its neck part by placing it upside down. That means the mouth of the Balloon and the bottle gets attached in opposite directions using their mouthparts. Now place the bottle set up in a container that consists of hot water in it. Leave it for some time. The Balloon starts inflating by filling its inside part with hot air molecules.
Bring the Balloon’s mouth part in an upside-down position over the neck part of the bottle. And then stretch the Balloon’s opening around the neck part of the bottle. But before that, you need to uncap the bottle. That’s it! Your Balloon’s opening nicely sits over the bottleneck part.
Boyle’s Law is valid at very high temperatures until or unless the gas remains as a gaseous matter. Because at high temperatures, the gases may change their state of mass, for which Boyle’s law is not applicable. Boyle’s law tells that the volume and pressure of a gas-related each other quite the opposite.
When you squeeze the bottle, the Balloon begins inflating itself because we squeeze some air molecules into it while squeezing the bottle. And due to more air occupying inside the Balloon, the Balloon starts expanding and inflates itself to fit the air molecules coming inside. When you stop squeezing the bottle, the balloon deflates.
When you let the Balloon warm up again, it starts inflating itself because of warmer air molecules. The warmer air molecules rise and enter into the Balloon, making it expand. Hot air molecules are less dense in weight and tend to travel upwards. And they require more space since they like to scatter in larger areas.
