This science experiment demonstration on triple point offers a deep insight into the world of the properties of water.
Have you ever noticed how ice floats at the top in oceans and many marine animals use to survive beneath the same?
This happens because water has a strange property that is quite different from most of the other substances! It’s solid-state (ice) is less dense than its liquid state because of which it floats and acts as an insulator to prevent the vast water bodies from freezing. Sounds strange!
Water has plenty of other properties that make it so different from other substances available on the Earth.
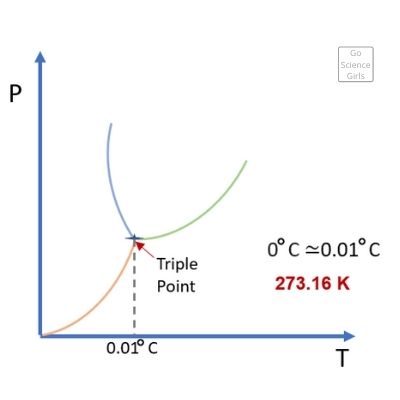
Do you know that any pure substance can exist in the solid, liquid, and gaseous state at the same time?
This sounds impossible, right? Well, this can happen, and the point of time at which this occurs is called the triple point.
You will be thrilled to know that pure water has a triple point at 0.01°C temperature, which is just above the freezing point.
The pressure at this point remains at 611.5 Pa. While the triple point temperature is known as T3, the triple point pressure is denoted by P3.
Now, why it is called the triple point?
Well, it is called so because it is at this point, a substance can coexist at all three states of matter, and all three states reach a state of thermodynamic equilibrium.
In the video demonstration, the same has been demonstrated.
Raw materials
- One large bell jar
- One petri dish
- Vacuum pump
- A small amount of cold water and ice
- A cork stand
- A combined system of temperature and pressure sensor
Triple Point of Water – Experiment Steps

- The petri dish is filled with ice-cold water (Do not fill it completely).
- Now, the halfway filled petri dish is placed inside the vacuum jar on a cork stand
- The combined system of temperature and pressure sensor between the bell jar and the pump is connected
- Next, the vacuum pump is started, and both temperatures as well as, pressure starts decreasing inside the jar
- Soon, the water started boiling!
Observation

When you conduct the experiment, you will see that at a certain point in time, water starts to boil.
You will see the small bubbles rising on the surface. However, after a few minutes, you will see that a thin layer of ice, but there is the real catch!
The water is still boiling, and this is just amazing! So, water is melting, freezing, and boiling all the three at the same time.
Now, you may be wondering how can this happen as you were decreasing the temperature and pressure? This may be because decreasing pressure on pure water increases its freezing point.
Why is the triple point important?
The triple point of water is significant because it is taken as the standard reference point for defining the Kelvin temperature scale.
The triple point of water is also used in sealed cells as pressure transfer standards.
The triple point of each pure substance is different. For example, mercury, it is at a temperature of −38.83440 °C and a pressure of 0.2 m Pa. Similarly, the triple point of CO2 lies at −56.60 °C and 517 kPa.
Just like there is the triple point of water, there also exists a critical point of water at which the liquid and the gaseous state of water become indistinguishable from each other.
Surprisingly, the temperature at which the water reaches the critical point is known as the critical temperature, and the pressure at which it reached the critical point is called critical pressure.
Resource
https://www.youtube.com/watch?v=Juz9pVVsmQQ&feature=emb_logo
https://ucscphysicsdemo.sites.ucsc.edu/physics-5b6b-demos/triple-point-of-water/
https://www.engineersedge.com/thermodynamics/triple_point.htm
